Abstract
Aim
Percutaneous left atrial appendage (LAA) closure has been established as alternative stroke prophylaxis in patients with non-valvular atrial fibrillation (AF) and high bleeding risk. However, little is known regarding the outcome after LAA closure depending on the HAS-BLED score.
Methods
A sub-analysis of the prospective, multicenter, Left-Atrium-Appendage Occluder Register—GErmany (LAARGE) registry was performed assessing three different groups with respect to the HAS-BLED score (0–2 [group 1] vs. 3–4 [group 2] vs. 5–7 [group 3]).
Results
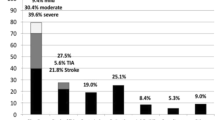
A total of 633 patients at 38 centers were enrolled. Of them, 9% (n = 59) were in group 1, 63% (n = 400) in group 2 and 28% (n = 174) in group 3. The Kaplan–Meier estimated 1-year composite of death, stroke and systemic embolism was 3.4% in group 1 vs. 10.4% in group 2 vs. 20.1% in group 3, respectively (p log-rank < 0.001). The difference was driven by death since stroke and systemic embolism did not show a significant difference between the groups. The rate of major bleeding at 1 year was 0% vs. 0% vs. 2.4%, respectively (p = 0.016).
Conclusion
The present data show that patients had similarly low rates of ischemic complications 1 year after LAA closure irrespective of the baseline bleeding risk. Higher HAS-BLED scores were associated with increased mortality due to higher age and more severe comorbidity of these patients.

Similar content being viewed by others
Abbreviations
- ACP:
-
Amplatzer cardiac plug
- INR:
-
International normalized ratio
- IQR:
-
Interquartile range
- LAA:
-
Left atrial appendage
- LAARGE:
-
Left-Atrium-Appendage Occluder Register—GErmany
- NOAK:
-
Novel oral anticoagulants
- TIA:
-
Transient ischemic attack
References
Reddy VY, Sievert H, Halperin J, Doshi SK, Buchbinder M, Neuzil P et al (2014) Percutaneous left atrial appendage closure vs warfarin for atrial fibrillation: a randomized clinical trial. JAMA 312:1988–1998
Brachmann J, Lewalter T, Akin I, Sievert H, Geist V, Zeymer U et al (2019) Interventional occlusion of left atrial appendage in patients with atrial fibrillation. Acute and long-term outcome of occluder implantation in the LAARGE Registry. J Interv Card Electrophysiol 2(4):403–412
Boersma LV, Schmidt B, Betts TR, Sievert H, Tamburino C, Teiger E et al (2016) Implant success and safety of left atrial appendage closure with the WATCHMAN device: peri-procedural outcomes from the EWOLUTION registry. Eur Heart J 37:2465–2474
Landmesser U, Tondo C, Camm J, Diener HC, Paul V, Schmidt B et al (2018) Left atrial appendage occlusion with the AMPLATZER Amulet device: one-year follow-up from the prospective global Amulet observational registry. EuroIntervention 14:e590–e597
Tzikas A, Holmes DR Jr, Gafoor S, Ruiz CE, Blomstrom-Lundqvist C, Diener HC et al (2016) Percutaneous left atrial appendage occlusion: the Munich consensus document on definitions, endpoints and data collection requirements for clinical studies. EuroIntervention 12:103–111
Price MJ, Reddy VY, Valderrabano M, Halperin JL, Gibson DN, Gordon N et al (2015) Bleeding outcomes after left atrial appendage closure compared with long-term warfarin: a pooled, patient-level analysis of the WATCHMAN randomized trial experience. JACC Cardiovasc Interv 8:1925–1932
Landmesser U, Schmidt B, Nielsen-Kudsk JE, Lam SCC, Park JW, Tarantini G et al (2017) Left atrial appendage occlusion with the AMPLATZER Amulet device: periprocedural and early clinical/echocardiographic data from a global prospective observational study. EuroIntervention 13:867–876
Reddy V, Price M, Valderrabano M, Sanchez J, Doshi S, Gordon N et al (2016) TCT-26 long-term event rates for left atrial appendage closure with WATCHMAN: PROTECT AF 5 Year and PREVAIL 3 year follow-up. J Am Coll Cardiol 68:B11
Boersma LV, Ince H, Kische S, Pokushalov E, Schmitz T, Schmidt B et al (2017) Efficacy and safety of left atrial appendage closure with WATCHMAN in patients with or without contraindication to oral anticoagulation: 1-year follow-up outcome data of the EWOLUTION trial. Heart Rhythm 14:1302–1308
Tzikas A, Shakir S, Gafoor S, Omran H, Berti S, Santoro G et al (2016) Left atrial appendage occlusion for stroke prevention in atrial fibrillation: multicentre experience with the AMPLATZER cardiac Plug. EuroIntervention 11:1170–1179
Ledwoch J, Sievert K, Boersma LVA, Bergmann MW, Ince H, Kische S et al (2020) Initial and long-term antithrombotic therapy after left atrial appendage closure with the WATCHMAN. Europace 22:1036–1043
Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY (2010) A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro heart survey. Chest 138:1093–1100
Bailey RD, Hart RG, Benavente O, Pearce LA (2001) Recurrent brain hemorrhage is more frequent than ischemic stroke after intracranial hemorrhage. Neurology 56:773–777
Vermeer SE, Algra A, Franke CL, Koudstaal PJ, Rinkel GJ (2002) Long-term prognosis after recovery from primary intracerebral hemorrhage. Neurology 59:205–209
Holmes DR Jr, Doshi SK, Kar S, Price MJ, Sanchez JM, Sievert H et al (2015) Left atrial appendage closure as an alternative to warfarin for stroke prevention in atrial fibrillation: a patient-level meta-analysis. J Am Coll Cardiol 65:2614–2623
Funding
The present Left-Atrium-Appendage Occluder Register—GErmany (LAARGE) was conducted independently from industry and only scientifically and financially sponsored by the Stiftung Institut für Herzinfarktforschung (IHF) Ludwigshafen, Germany. For the biometrical analyses of the present work, the IHF was financially supported by an unrestricted grant from Boston Scientific.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Jakob Ledwoch: none.
Jennifer Franke: none.
Johannes Brachmann: none.
Thorsten Lewalter: Speaker honorary from St. Jude Medical/Abbott Vascular and Boston Scientific.
Ibrahim Akin: none.
Jochen Senges: none.
Matthias Hochadel: none.
Christian Weiß: none.
Alexander Krapivsky: none.
Horst Sievert: Study honorary, travel expenses, consulting fees from Access Closure, AGA, Angiomed, Ardian, Arstasis, Atritech, Atrium, Avinger, Bard, Boston Scientific, Bridgepoint, CardioKinetix, CardioMEMS, Coherex, Contego, CSI, EndoCross, EndoTex, Epitek, Evalve, ev3, FlowCardia, Gore, Guidant, Lumen Biomedical, HLT, Kensey Nash, Kyoto Medical, Lifetech, Lutonix, Medinol, Medtronic, NDC, NMT, OAS, Occlutech, Osprey, Ovalis, Pathway, PendraCare, Percardia, pfm Medical, Recor, Rox Medical, Sadra, Sorin, Spectranetics, SquareOne, Trireme, Trivascular, Viacor, Velocimed, Veryan.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ledwoch, J., Franke, J., Brachmann, J. et al. Impact of HAS-BLED Score on outcome after percutaneous left atrial appendage closure: insights from the German Left Atrial Appendage Occluder Registry LAARGE. Clin Res Cardiol 111, 541–547 (2022). https://doi.org/10.1007/s00392-021-01911-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-021-01911-1