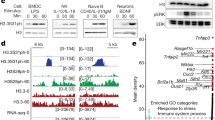
Abstract
The histone deacetylases (HDACs) are a superfamily of chromatin-modifying enzymes that silence transcription through the modification of histones. Among them, HDAC3 is unique in that interaction with nuclear receptor corepressors 1 and 2 (NCoR1/2) is required to engage its catalytic activity1,2,3. However, global loss of HDAC3 also results in the repression of transcription, the mechanism of which is currently unclear4,5,6,7,8. Here we report that, during the activation of macrophages by lipopolysaccharides, HDAC3 is recruited to activating transcription factor 2 (ATF2)-bound sites without NCoR1/2 and activates the expression of inflammatory genes through a non-canonical mechanism. By contrast, the deacetylase activity of HDAC3 is selectively engaged at ATF3-bound sites that suppress Toll-like receptor signalling. Loss of HDAC3 in macrophages safeguards mice from lethal exposure to lipopolysaccharides, but this protection is not conferred upon genetic or pharmacological abolition of the catalytic activity of HDAC3. Our findings show that HDAC3 is a dichotomous transcriptional activator and repressor, with a non-canonical deacetylase-independent function that is vital for the innate immune system.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All sequencing data from RNA-seq, ChIP–seq and GRO-seq analyses have been deposited to the Gene Expression Omnibus under accession number GSE140611. ChIP–seq data for Fos, JunB, JunD and CREB in LPS-stimulated BMDMs were obtained from GSE99895. All other data are available from the corresponding author upon reasonable request.
References
Guenther, M. G., Barak, O. & Lazar, M. A. The SMRT and N-CoR corepressors are activating cofactors for histone deacetylase 3. Mol. Cell. Biol. 21, 6091–6101 (2001).
Watson, P. J., Fairall, L., Santos, G. M. & Schwabe, J. W. R. Structure of HDAC3 bound to co-repressor and inositol tetraphosphate. Nature 481, 335–340 (2012).
You, S. H. et al. Nuclear receptor co-repressors are required for the histone-deacetylase activity of HDAC3 in vivo. Nat. Struct. Mol. Biol. 20, 182–187 (2013).
Chen, X. et al. Requirement for the histone deacetylase Hdac3 for the inflammatory gene expression program in macrophages. Proc. Natl Acad. Sci. USA 109, E2865–E2874 (2012).
Mullican, S. E. et al. Histone deacetylase 3 is an epigenomic brake in macrophage alternative activation. Genes Dev. 25, 2480–2488 (2011).
Greer, C. B. et al. Histone deacetylases positively regulate transcription through the elongation machinery. Cell Rep. 13, 1444–1455 (2015).
Lewandowski, S. L., Janardhan, H. P. & Trivedi, C. M. Histone deacetylase 3 coordinates deacetylase-independent epigenetic silencing of transforming growth factor- β1 (TGF-β1) to orchestrate second heart field development. J. Biol. Chem. 290, 27067–27089 (2015).
Emmett, M. J. et al. Histone deacetylase 3 prepares brown adipose tissue for acute thermogenic challenge. Nature 546, 544–548 (2017).
Mann, B. S., Johnson, J. R., Cohen, M. H., Justice, R. & Pazdur, R. FDA approval summary: vorinostat for treatment of advanced primary cutaneous T-cell lymphoma. Oncologist 12, 1247–1252 (2007).
Gryder, B. E., Sodji, Q. H. & Oyelere, A. K. Targeted cancer therapy: giving histone deacetylase inhibitors all they need to succeed. Future Med. Chem. 4, 505–524 (2012).
De Souza, C. & Chatterji, B. P. HDAC inhibitors as novel anti-cancer therapeutics. Recent Pat. Anticancer Drug Discov. 10, 145–162 (2015).
Kazantsev, A. G., & Leslie, M. T. Therapeutic application of histone deacetylase inhibitors for central nervous system disorders. Nat. Rev. Drug Discov. 10, 854–868 (2008).
Leoni, F. et al. The antitumor histone deacetylase inhibitor suberoylanilide hydroxamic acid exhibits antiinflammatory properties via suppression of cytokines. Proc. Natl Acad. Sci. USA 99, 2995–3000 (2002).
Sun, Z. et al. Deacetylase-independent function of HDAC3 in transcription and metabolism requires nuclear receptor corepressor. Mol. Cell 52, 769–782 (2013).
Marinelli, C. et al. Ligand engagement of Toll-like receptors regulates their expression in cortical microglia and astrocytes. J. Neuroinflammation 12, 244 (2015).
Gasperini, M. et al. A genome-wide framework for mapping gene regulation via cellular genetic screens. Cell 176, 377–390.e19 (2019).
Fonseca, G. J. et al. Diverse motif ensembles specify non-redundant DNA binding activities of AP-1 family members in macrophages. Nat. Commun. 10, 414 (2019).
Barish, G. D. et al. Bcl-6 and NF-kappaB cistromes mediate opposing regulation of the innate immune response. Genes Dev. 24, 2760–2765 (2010).
Chen, S. et al. Valproic acid attenuates traumatic spinal cord injury-induced inflammation via STAT1 and NF-κB pathway dependent of HDAC3. J. Neuroinflammation 15, 150 (2018).
Ziesché, E. et al. The coactivator role of histone deacetylase 3 in IL-1-signaling involves deacetylation of p65 NF-κB. Nucleic Acids Res. 41, 90–109 (2012).
Chen, L.-f., Fischle, W., Verdin, E. & Greene, W. C. Duration of nuclear NF-κB action regulated by reversible acetylation. Science 293, 1653–1657 (2001).
Kiernan, R. et al. Post-activation turn-off of NF-κ B-dependent transcription is regulated by acetylation of p65. J. Biol. Chem. 278, 2758–2766 (2003).
Khuu, C. H., Barrozo, R. M., Hai, T. & Weinstein, S. L. Activating transcription factor 3 (ATF3) represses the expression of CCL4 in murine macrophages. Mol. Immunol. 44, 1598–1605 (2007).
De Nardo, D. et al. High-density lipoprotein mediates anti-inflammatory reprogramming of macrophages via the transcriptional regulator ATF3. Nat. Immunol. 15, 152 (2014).
Luo, H. et al. ATF3 inhibits tenascin-C-induced foam cell formation in LPS-stimulated THP-1 macrophages by suppressing TLR-4. J. Atheroscler. Thromb. 22, 1214–1223 (2015).
Bambouskova, M. et al. Electrophilic properties of itaconate and derivatives regulate the IκBζ–ATF3 inflammatory axis. Nature 556, 501–504 (2018).
Labzin, L. I. et al. ATF3 is a key regulator of macrophage IFN responses. J. Immunol. 195, 4446–4455 (2015).
Yan, Q. et al. Nuclear factor-κB binding motifs specify Toll-like receptor-induced gene repression through an inducible repressosome. Proc. Natl Acad. Sci. USA 109, 14140–14145 (2012).
Copeland, S., Warren, H. S., Lowry, S. F., Calvano, S. E. & Remick, D. Acute inflammatory response to endotoxin in mice and humans. Clin. Diagn. Lab. Immunol. 12, 60–67 (2005).
Honore, P. M. et al. Cytokine removal in human septic shock: where are we and where are we going? Ann. Intensive Care 9, 56 (2019).
Wheeler, D. S., et al. Induction of endotoxin tolerance enhances bacterial clearance and survival in murine polymicrobial sepsis. Shock 30, 267–273 (2008).
Roger, T. et al. Histone deacetylase inhibitors impair innate immune responses to Toll-like receptor agonists and to infection. Blood 117, 1205–1217 (2011).
Lin, H.-S. et al. Anti-rheumatic activities of histone deacetylase (HDAC) inhibitors in vivo in collagen-induced arthritis in rodents. Br. J. Pharmacol. 150, 862–872 (2007).
Hu, Q. & Mao, D. Histone deacetylase inhibitor SAHA attenuates post-seizure hippocampal microglia TLR4/MYD88 signaling and inhibits TLR4 gene expression via histone acetylation. BMC Neurosci. 17, 22 (2016).
Huber, K. et al. Inhibitors of histone deacetylases: correlation between isoform specificity and reactivation of HIV type 1 (HIV-1) from latently infected cells. J. Biol. Chem. 286, 22211–22218 (2011).
Kuang, Z. et al. The intestinal microbiota programs diurnal rhythms in host metabolism through histone deacetylase 3. Science 365, 1428–1434 (2019).
Lee, M. S. et al. Genome-wide profiling of in vivo LPS-responsive genes in splenic myeloid cells. Mol. Cells 35, 498–513 (2013).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Chen, E. Y. et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinformatics 14, 128 (2013).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. 102, 15545–15550 (2005).
Mootha, V. K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 34, 267–273 (2003).
Wang, D., et al. Reprogramming transcription by distinct classes of enhancers functionally defined by eRNA. Nature 474, 390–394 (2011).
Kaikkonen, M. U. et al. Remodeling of the enhancer landscape during macrophage activation is coupled to enhancer transcription. Mol. Cell 51, 310–325 (2013).
Fang, B. et al. Circadian enhancers coordinate multiple phases of rhythmic gene transcription in vivo. Cell 159, 1140–1152 (2014).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Heinz, S. et al. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 38, 576–589 (2010).
Robinson, J. T. et al. Integrative genomics viewer. Nat. Biotechnol. 29, 24–26 (2011).
Acknowledgements
We thank D. Steger and H. Goodarzi for discussions, J. Marinis and J. DiSpirito for help with setting up the macrophage system, and the Functional Genomics Core of the Penn Diabetes Research Center (National Institutes of Health (NIH) P30 19525) for next-generation sequencing. This work was supported by NIH R01 DK43806 (M.A.L.), NIH T32 DK07314 (A.K.H.), American Diabetes Association 1-18-PDF-126 (M.A.) and the JPB Foundation.
Author information
Authors and Affiliations
Contributions
H.C.B.N. and M.A.L. conceived the project, designed experiments, analysed results and wrote the manuscript; H.C.B.N. performed animal experiments and LPS susceptibility assays on genetic mouse models, tissue culture, HDAC3/ATF2/ATF3/p65 immunoblots, RNA-seq, HDAC3/ATF2/NCor1/NCoR2 ChIP–seq, and GRO-seq experiments, as well as bioinformatic analyses. M.A. performed isolation of peritoneal macrophages from the LPS-injected genetic mouse model for RNA-seq/ChIP–seq analyses, and the LPS susceptibility assay on C57BL/6, control and MHD3KO mice treated with different doses of SAHA. A.K.H. performed the H3K27Ac immunoblot, and ChIP–seq analyses for H3K27Ac, ATF3 and p65.
Corresponding author
Ethics declarations
Competing interests
M.A.L. receives research support from Pfizer for unrelated work, serves as an advisory board member for Pfizer, has consulted for Novartis, Madrigal, Calico and Third Rock, and holds equity in KDAC Therapeutics.
Additional information
Peer review information Nature thanks Gioacchino Natoli, Inez Rogatsky and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 DA-independent and DA-dependent functions of HDAC3 in the inflammatory response to LPS.
a, Scatter plots of RNA-seq experiments in control, MHD3KO, HDAC3(Y298F) and WT-rescue BMDM, with or without LPS, showing correlation between biological replicates using either log2 transformation (log2(x+1)), variance stabilizing transformation (vst), or regularized-logarithm transformation (rlog) of normalized read counts. b, Principal component analysis (PCA) plot using the vst values from RNA-seq experiments in control, MHD3KO, HDAC3(Y298F) and WT-rescue BMDM, with or without LPS (n = 4 biological replicates). c, Heat map of sample-to-sample distances using the vst values from RNA-seq experiments in control, MHD3KO, HDAC3(Y298F) and WT-rescue BMDM, with or without LPS (n = 4 biological replicates). d, Empirical cumulative distribution function (CDF) and associated two-sided Kolmogorov–Smirnov test D statistics of LPS-stimulated changes in statistically modelled DA-independent gene expression (385 genes) for control, MHD3KO, HDAC3(Y298F) and WT-rescue BMDM (n = 4 biological replicates). e, Empirical CDF and associated two-sided Kolmogorov–Smirnov test D statistics of LPS-stimulated changes in statistically modelled DA-dependent gene expression (377 genes) for control, MHD3KO, HDAC3(Y298F) and WT-rescue BMDM (n = 4 biological replicates). f, Heat map showing 142 LPS-downregulated genes that were rescued by wild-type HDAC3 but not by HDAC3(Y298F) (DA-dependent genes: n = 4 biological replicates, DE cutoff: |log2FC| > 1, two-sided Benjamini–Hochberg-adjusted FDR < 0.05 as determined by edgeR likelihood ratio test). g, Gene ontology analysis of 142 LPS-downregulated genes (n = 4 biological replicates). q-values represent Benjamini–Hochberg-adjusted one-sided hypergeometric P values for over-representation as determined by Enrichr. h, LPS-stimulated (762 genes) and IL4-stimulated (405 genes) transcriptomic correlation of MHD3KO and HDAC3(Y298F), compared with control BMDM (n = 4 biological replicates). Heat map showing Spearman’s correlation ρ values with corresponding two-sided P values calculated by t-tests.
Extended Data Fig. 2 Differential recruitment and enhancer activity of HDAC3 at LPS-responsive genes.
a, Scatter plots of ChIP–seq experiments for HDAC3 and H3K27Ac in vehicle or LPS-treated control BMDM (n = 3 biological replicates) showing correlation between biological replicates using tag counts per base-pair (TPB) at identified peak regions. Pearson’s P values were calculated with two-sided t-tests. b, Average density profiles in RPM of HDAC3 ChIP–seq showing mean ± s.e.m. (n = 3 biological replicates) at all identified HDAC3 peaks (10,966 total) in control and MHD3KO macrophages, with or without LPS. c, Average density profiles in RPM of GRO-seq showing mean ± s.e.m. (n = 3 biological replicates) at all identified eRNAs (12,192 total) in control and MHD3KO BMDM, with or without LPS. d, Average density profiles in RPM of H3K27Ac showing mean ± s.e.m. (n = 3 biological replicates) at all identified H3K27Ac peaks (50,247 total) in control and MHD3KO macrophages, with or without LPS. e, Western blot (performed independently twice) of HDAC3 and H3K27Ac protein levels in control and MHD3KO macrophages, with vinculin and histone H3 as loading controls. f, g, Genome-browser tracks showing three biologically replicated examples of enhancer and gene body activity as measured by GRO-seq relative to HDAC3 ChIP–seq peaks in control and MHD3KO macrophages, with or without LPS near DA-independent (f) or DA-dependent (g) genes. h, Top de novo enriched motifs at HDAC3-bound genomic regions (10,966 sites from n = 3 biological replicates). Statistics were determined by HOMER with one-sided hypergeometric P values for over-representation.
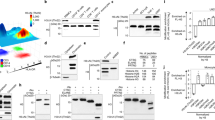
Extended Data Fig. 3 ATF2 and ATF3 differentially mediate HDAC3 transcriptional effects at DA-independent and DA-dependent sites, respectively.
a, Comparison of the performance of TBA classifiers modelled against DA-independent and DA-dependent 200-bp-DNA sequences as measured by the area under the receiver operating characteristic curve (auROC, n = 5 independent train-test iterations, data shown as mean ± s.d.). b, Heat map showing relative enrichment of several AP-1 family member DNA motifs as determined by TBA with likelihood ratio test against DA-independent and DA-dependent 200-bp-DNA sequences across 5 train-test iterations. The non-redundant, merged motifs from TBA include ATF1, 4, 5, 6, 7, Jun family, Fos family, and other related bZIP factors. c, Average density profiles in RPM of Fos, JunB, JunD and CREB ChIP–seqs obtained from Gene Expression Omnibus database at HDAC3-bound sites near either DA-independent or DA-dependent genes in LPS-treated BMDM. d, Average density profiles in RPM of bidirectional eRNA transcription measured by GRO-seq showing mean ± s.e.m. (n = 3 biological replicates) at HDAC3-bound enhancers with ATF2 motif (1,680 sites) or ATF3 motif (3,673 sites) (two-sided Wilcoxon’s P = 8.6 × 10−127) in LPS-treated BMDM. e, f, Genome-browser tracks showing HDAC3, ATF2 and ATF3 ChIP–seq peaks at enhancer elements upstream of Clec2d (e) or Gas6 (f), as well as gene body activity as measured by RNA-seq in control, MHD3KO, HDAC3(Y298F) and WT rescue macrophages, with or without LPS. g, h, Dual-luciferase assays of transcription driven by the Clec2d (g) or Gas6 (h) enhancers in control, MHD3KO, HDAC3(Y298F) rescue, WT-rescue (WT), ATF2-depleted and ATF3-depleted BMDM, with or without LPS stimulation. Data shown are mean ± s.d., n = 4 biological replicates. P values were calculated by one-way ANOVA. DA-independent sites = 172, DA-dependent sites = 141.
Extended Data Fig. 4 ATF2 and ATF3 recruit HDAC3 to sites near DA-independent and DA-dependent genes, respectively.
a, Scatter plots of ChIP–seq experiments for ATF2, ATF3, NCoR1, NCoR2 and p65 in LPS-treated BMDM (n = 3 biological replicates) showing correlation between biological replicates using average tag counts per base-pair (TPB) at identified peak regions. Pearson’s P values were calculated with two-sided t-tests. b, Relative gene expression levels of Atf2, Atf3 and p65 (also known as Rela) as measured by qPCR in control (siC), ATF2-depleted (siAtf2), ATF3-depleted (siAtf3), or p65-depleted (sip65) BMDM (n = 3 biological replicates). Data shown mean ± s.d., P values calculated by two-sided Student’s t-test. c, Western blots (performed once) of ATF2, ATF3 and p65 protein levels with vinculin as loading control for control (siControl), ATF2-depleted (siAtf2), ATF3-depleted (siAtf3) or p65-depleted (sip65) BMDM, each with 3 independent siRNAs. d, Heat map of sample-to-sample distances using the vst values from RNA-seq experiments in control (siC), ATF2-depleted (siAtf2), ATF3-depleted (siAtf3) or p65-depleted (sip65) BMDM, with or without LPS (n = 3). e, f, Genome-browser tracks showing biologically replicated examples of HDAC3 ChIP–seq peaks in control (siControl), ATF2-depleted (siAtf2), ATF3-depleted (siAtf3) or p65-depleted (sip65) LPS-stimulated BMDM near DA-independent (e) or DA-dependent (f) genes. g, h, Average density profiles in RPM of p65 ChIP–seq showing mean ± s.e.m. (n = 3 biological replicates) at HDAC3-bound sites near DA-independent genes (Control LPS versus MHD3KO LPS two-sided Wilcoxon’s P = 0.063) (g) or DA-dependent genes (Control LPS versus MHD3KO LPS two-sided Wilcoxon’s P = 2.4 × 10−16) in control and MHD3KO BMDM, with or without LPS (h). i, j, Box-and-whisker plot showing minimum, maximum, median, first quartile and third quartile from quantification of average ChIP–seq signal (n = 3 biological replicates) in RPM for input, HDAC3, NCoR1 and NCoR2 at HDAC3-bound sites near DA-independent (i) or DA-dependent (j) genes. P values were calculated with two-sided Mann–Whitney test. k, l, Average density profiles in RPM showing mean ± s.e.m. (n = 3 biological replicates) of NCoR1 (two-sided Wilcoxon’s P = 4.7 × 10−4) (k) and NCoR2 (two-sided Wilcoxon’s P = 1.2 × 10−5) (l) genomic colocalization with either ATF2 (19,594 peaks) or ATF3 (57,041 peaks). m, n, Genome-browser tracks showing three biologically replicated examples of ChIP–seq peaks for HDAC3, ATF2, ATF3, NCoR1, and NCoR2 in LPS-stimulated BMDM near DA-independent (k) or DA-dependent (l) genes. DA-independent sites = 172, DA-dependent sites = 141.
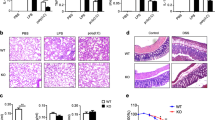
Extended Data Fig. 5 Loss of HDAC3 protein but not deacetylase activity protects mice from acute endotoxic shock.
a, Kaplan–Meier curves of wild-type C57BL/6 mice injected with increasing doses of LPS and observed for 120 h (n = 4 independent mice). b, Serum cytokine concentrations measured by ELISA for IL6 and TNF in 10 mg kg−1 LPS-injected control, MHD3KO, NSDAD, C57BL/6 mice (n = 5 independent mice) administered with increasing doses of SAHA (25, 100, 400 mg kg−1), and untreated control (n = 3 independent mice). Data shown mean ± s.d. P values calculated by one-way ANOVA. c, PCA plot using the vst values from RNA-seq experiments of in vivo peritoneal macrophages from control, MHD3KO and NSDAD mice injected with vehicle control (PBS) or 10 mg kg−1 LPS (n = 4 biological replicates, except for KO vehicle and NSDAD LPS with n = 3 biological replicates). d, Scatter plot showing correlation between in vitro (n = 4 biological replicates) and in vivo (n = 3 biological replicates) DA-independent differential gene expression (174 genes). Pearson’s P value was calculated with two-sided t-test. e, Empirical CDF and associated two-sided Kolmogorov–Smirnov test D statistics of LPS-stimulated changes in statistically modelled DA-independent in vivo gene expression (251 genes) for peritoneal macrophages from control (n = 4 biological replicates), MHD3KO (n = 4 biological replicates) and NSDAD (n = 3 biological replicates). f, Heat map showing 177 LPS-downregulated, HDAC3-dependent differentially expressed genes in vivo that were not rescued by NSDAD (DA-dependent genes: n = 4 biological replicates, except for KO vehicle and NSDAD LPS with n = 3 biological replicates; DE cutoff: |log2FC| > 1, two-sided Benjamini–Hochberg-adjusted FDR < 0.05 as determined by edgeR likelihood ratio test). g, Empirical CDF and associated two-sided Kolmogorov–Smirnov test D statistics of LPS-stimulated changes in statistically modelled DA-dependent in vivo gene expression (404 genes) for peritoneal macrophages from control (n = 4 biological replicates), MHD3KO (n = 4 biological replicates) and NSDAD (n = 3 biological replicates). h, Gene set enrichment analysis (GSEA) for the in vivo DA-dependent transcriptome (177 genes) showing enrichment of KEGG Toll-like receptor signalling pathway that was more upregulated in peritoneal macrophages from LPS-treated NSDAD mice (n = 3 biological replicates) than from LPS-treated control mice (n = 4 biological replicates). Bar graph showing Tlr4 gene expression level in normalized counts of peritoneal macrophages from control and NSDAD treated with LPS. Data shown mean ± s.d., two-sided P value was calculated by Student’s t-test. i, j, Average density profiles in RPM of HDAC3 ChIP–seq showing mean ± s.e.m. (n = 3 biological replicates) at HDAC3-bound sites near 167 in vivo DA-independent genes (two-sided Wilcoxon’s P = 1.7 × 10−23) (i) or 165 DA-dependent genes (two-sided Wilcoxon’s P = 0.153) (j) in peritoneal macrophages from vehicle- or LPS-treated control mice.
Extended Data Fig. 6 Dose-dependent effects of HDAC inhibitor SAHA on endotoxin susceptibility.
a, Kaplan–Meier curves of C57BL/6 mice subjected to 10 mg kg−1 of intraperitoneal injection of purified LPS, with increasing doses of SAHA (25, 100, 400 mg kg−1). SAHA vehicle (veh), 10% DMSO in PBS. *P = 0.036, **P = 0.001, ***P = 0.00065, n = 10 independent mice, calculated by two-sided Mantel–Cox test. b, Heat map showing LPS-induced transcriptional changes as a function of log2(fold change) (log2FC) of 179 in vivo DA-independent genes in LPS-exposed BMDM treated with increasing dose of SAHA (25, 100, 400 nM, n = 3 biological replicates) and peritoneal macrophages from LPS-exposed MHD3KO (n = 4 biological replicates) and NSDAD mice (n = 3 biological replicates). c, Average density profiles in RPM of HDAC3 ChIP–seq showing means of 3 biological replicates at HDAC3-bound sites near 172 DA-independent genes. d, Genome-browser tracks showing 3 biologically replicated examples of ChIP–seq peaks for HDAC3 in LPS-stimulated BMDM treated with increasing dose of SAHA (25, 100, 400 nM) near DA-independent genes. e, Average density profiles in RPM of HDAC3 ChIP–seq showing means of 3 biological replicates at HDAC3-bound sites near 141 DA-dependent genes. f, Genome-browser tracks showing 3 biologically replicated examples of ChIP–seq peaks for HDAC3 in LPS-stimulated BMDM treated with increasing dose of SAHA (25, 100, 400 nM) near DA-dependent genes.
Supplementary information
Supplementary Information
This file contains Supplementary Figure 1. Raw images of blots. Unprocessed images of scanned immunoblot membranes for data shown in Fig. 1b, Extended Data Fig. 2e, and Extended Data Fig. 4c. Supplementary Table 1. List of siRNAs, Ultramers, primers, and antibodies. The table include sequences for IDT siRNAs against Atf2, Atf3, and Rela, IDT ultramers for luciferase assays, primers used for qRT-PCR experiments, as well as antibodies used for immunoprecipitation and Western Blot experiments. Supplementary Table 2. ChIP-seq quality controls reporting the number of mapped reads, number of identified peaks, and corresponding FrIP scores. Supplementary Tables 3a-f. Raw output of de novo motif analyses as performed by HOMER for all performed ChIP-seqs: HDAC3, ATF2, ATF3, p65, NCoR1, and NCoR2. Statistics reporting one-sided hypergeometric unadjusted p-values calculated by HOMER for over-representation or under-representation of target DNA sequence over background DNA sequences.
Rights and permissions
About this article
Cite this article
Nguyen, H.C.B., Adlanmerini, M., Hauck, A.K. et al. Dichotomous engagement of HDAC3 activity governs inflammatory responses. Nature 584, 286–290 (2020). https://doi.org/10.1038/s41586-020-2576-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2576-2
This article is cited by
-
Nuclear receptor corepressors non-canonically drive glucocorticoid receptor-dependent activation of hepatic gluconeogenesis
Nature Metabolism (2024)
-
Integrative function of histone deacetylase 3 in inflammation
Molecular Biology Reports (2024)
-
Targeting HDAC3 to overcome the resistance to ATRA or arsenic in acute promyelocytic leukemia through ubiquitination and degradation of PML-RARα
Cell Death & Differentiation (2023)
-
The role of HDAC3 and its inhibitors in regulation of oxidative stress and chronic diseases
Cell Death Discovery (2023)
-
Histone demethylase KDM5B licenses macrophage-mediated inflammatory responses by repressing Nfkbia transcription
Cell Death & Differentiation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.