Abstract
Background
Scarce data exist about early infective endocarditis (IE) after trans-catheter aortic valve replacement (TAVR).
Objective
The objective was to evaluate the characteristics, management, and outcomes of very early (VE) IE (≤ 30 days) after TAVR.
Methods
This multicenter study included a total of 579 patients from the Infectious Endocarditis after TAVR International Registry who had the diagnosis of definite IE following TAVR.
Results
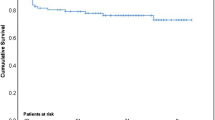
Ninety-one patients (15.7%) had VE-IE. Factors associated with VE-IE (vs. delayed IE (D-IE)) were female gender (p = 0.047), the use of self-expanding valves (p < 0.001), stroke (p = 0.019), and sepsis (p < 0.001) after TAVR. Staphylococcus aureus was the main pathogen among VE-IE patients (35.2% vs. 22.7% in the D-IE group, p = 0.012), and 31.2% of Staphylococcus aureus infections in the VE-IE group were methicillin-resistant (vs. 14.3% in the D-IE group, p = 0.001). The second-most common germ was enterococci (34.1% vs. 24.4% in D-IE cases, p = 0.05). VE-IE was associated with very high in-hospital (44%) and 1-year (54%) mortality rates. Acute renal failure following TAVR (p = 0.001) and the presence of a non-enterococci pathogen (p < 0.001) were associated with an increased risk of death.
Conclusion
A significant proportion of IE episodes following TAVR occurs within a few weeks following the procedure and are associated with dismal outcomes. Some baseline and TAVR procedural factors were associated with VE-IE, and Staphylococcus aureus and enterococci were the main causative pathogens. These results may help to select the more appropriate antibiotic prophylaxis in TAVR procedures and guide the initial antibiotic therapy in those cases with a clinical suspicion of IE.
Graphical abstract
Very early infective endocarditis after trans-catheter aortic valve replacement. VE-IE indicates very early infective endocarditis (≤30 days post TAVR). D-IE indicates delayed infective endocarditis.




Similar content being viewed by others
Abbreviations
- D-IE:
-
Delayed infective endocarditis
- IE:
-
Infective endocarditis
- IQR:
-
Interquartile range
- PVE:
-
Prosthetic valve endocarditis
- TAVR:
-
Trans-catheter aortic valve replacement
- THV:
-
Trans-catheter heart valve
- VE-IE:
-
Very early infective endocarditis
References
Regueiro A, Linke A, Latib A et al (2016) Association between transcatheter aortic valve replacement and subsequent infective endocarditis and in-hospital death. JAMA 316:1083–1092. https://doi.org/10.1001/jama.2016.12347
Ando T, Ashraf S, Villablanca PA et al (2019) Meta-analysis comparing the incidence of infective endocarditis following transcatheter aortic valve implantation versus surgical aortic valve replacement. Am J Cardiol 123:827–832. https://doi.org/10.1016/j.amjcard.2018.11.031
Fauchier L, Bisson A, Herbert J et al (2020) Incidence and outcomes of infective endocarditis after transcatheter aortic valve implantation versus surgical aortic valve replacement. Clin Microbiol Infect 26:1368–1374. https://doi.org/10.1016/j.cmi.2020.01.036
Summers MR, Leon MB, Smith CR et al (2019) Prosthetic valve endocarditis after TAVR and SAVR: insights from the PARTNER trials. Circulation 140:1984–1994. https://doi.org/10.1161/CIRCULATIONAHA.119.041399
Butt JH, Ihlemann N, De Backer O et al (2019) Long-term risk of infective endocarditis after transcatheter aortic valve replacement. J Am Coll Cardiol 73:1646–1655. https://doi.org/10.1016/j.jacc.2018.12.078
Stortecky S, Heg D, Tueller D et al (2020) Infective endocarditis after transcatheter aortic valve replacement. J Am Coll Cardiol 75:3020–3030. https://doi.org/10.1016/j.jacc.2020.04.044
Habib G, Lancellotti P, Antunes MJ et al (2015) 2015 ESC Guidelines for the management of infective endocarditis. the task force for the management of infective endocarditis of the European Society of Cardiology (ESC)Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J 36:3075–3128. https://doi.org/10.1093/eurheartj/ehv319
Kappetein AP, Head SJ, Généreux P et al (2012) Updated standardized endpoint definitions for transcatheter aortic valve implantation: the Valve Academic Research Consortium-2 consensus document. J Am Coll Cardiol 60:1438–1454. https://doi.org/10.1016/j.jacc.2012.09.001
Mehta RL, Kellum JA, Shah SV et al (2007) Acute Kidney Injury Network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 11:R31. https://doi.org/10.1186/cc5713
Siciliano RF, Randi BA, Gualandro DM et al (2018) Early-onset prosthetic valve endocarditis definition revisited: prospective study and literature review. Int J Infect Dis 67:3–6. https://doi.org/10.1016/j.ijid.2017.09.004
Benito N, Miró JM, de Lazzari E et al (2009) Health care-associated native valve endocarditis: importance of non-nosocomial acquisition. Ann Intern Med 150:586–594. https://doi.org/10.7326/0003-4819-150-9-200905050-00004
Fernández-Hidalgo N, Almirante B, Tornos P et al (2008) Contemporary epidemiology and prognosis of health care-associated infective endocarditis. Clin Infect Dis 47:1287–1297. https://doi.org/10.1086/592576
Graffunder EM, Venezia RA (2002) Risk factors associated with nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infection including previous use of antimicrobials. J Antimicrob Chemother 49:999–1005. https://doi.org/10.1093/jac/dkf009
Roghmann M-C, Lydecker AD, Hittle L et al (2017) Comparison of the microbiota of older adults living in nursing homes and the community. mSphere 2:e00210-17. https://doi.org/10.1128/mSphere.00210-17
Bonten MJ, Hayden MK, Nathan C et al (1996) Epidemiology of colonisation of patients and environment with vancomycin-resistant enterococci. The Lancet 348:1615–1619. https://doi.org/10.1016/S0140-6736(96)02331-8
Miller WR, Munita JM, Arias CA (2014) Mechanisms of antibiotic resistance in enterococci. Expert Rev Anti Infect Ther 12:1221–1236. https://doi.org/10.1586/14787210.2014.956092
Chambers HF, DeLeo FR (2009) Waves of resistance: Staphylococcus aureus in the Antibiotic Era. Nat Rev Microbiol 7:629–641. https://doi.org/10.1038/nrmicro2200
Engelman R, Shahian D, Shemin R, et al (2007) The Society of Thoracic Surgeons Practice Guideline Series: Antibiotic Prophylaxis in Cardiac Surgery, Part II: Antibiotic Choice. Ann Thorac Surg 83:1569–1576. https://doi.org/10.1016/j.athoracsur.2006.09.046
Finkelstein R, Rabino G, Mashiah T et al (2002) Vancomycin versus cefazolin prophylaxis for cardiac surgery in the setting of a high prevalence of methicillin-resistant staphylococcal infections. J Thorac Cardiovasc Surg 123:326–332. https://doi.org/10.1067/mtc.2002.119698
Chenoweth CE, DePestel DD, Prager RL (2005) Are cephalosporins adequate for antimicrobial prophylaxis for cardiac surgery involving implants? Clin Infect Dis 41:122–123. https://doi.org/10.1086/430831 (author reply 123-124)
Baddour LM, Epstein AE, Erickson CC et al (2010) Update on cardiovascular implantable electronic device infections and their management: a scientific statement from the American Heart Association. Circulation 121:458–477. https://doi.org/10.1161/CIRCULATIONAHA.109.192665
Conen A, Stortecky S, Moreillon P et al (2021) A review of recommendations for infective endocarditis prevention in patients undergoing transcatheter aortic valve implantation. EuroIntervention 16:1135–1140. https://doi.org/10.4244/EIJ-D-19-00993
Kachroo S, Dao T, Zabaneh F et al (2006) Tolerance of vancomycin for surgical prophylaxis in patients undergoing cardiac surgery and incidence of vancomycin-resistant enterococcus colonization. Ann Pharmacother 40:381–385. https://doi.org/10.1345/aph.1G565
Sambola A, Fernández-Hidalgo N, Almirante B et al (2010) Sex differences in native-valve infective endocarditis in a single tertiary-care hospital. Am J Cardiol 106:92–98. https://doi.org/10.1016/j.amjcard.2010.02.019
Aksoy O, Meyer LT, Cabell CH et al (2007) Gender differences in infective endocarditis: pre- and co-morbid conditions lead to different management and outcomes in female patients. Scand J Infect Dis 39:101–107. https://doi.org/10.1080/00365540600993285
Chandrasekhar J, Dangas G, Yu J et al (2016) Sex-based differences in outcomes with transcatheter aortic valve therapy: TVT registry from 2011 to 2014. J Am Coll Cardiol 68:2733–2744. https://doi.org/10.1016/j.jacc.2016.10.041
Wang HE, Gamboa C, Warnock DG, Muntner P (2011) Chronic kidney disease and risk of death from infection. Am J Nephrol 34:330–336. https://doi.org/10.1159/000330673
Murdoch DR, Corey GR, Hoen B et al (2009) Clinical presentation, etiology, and outcome of infective endocarditis in the 21st century: the International Collaboration on Endocarditis-Prospective Cohort Study. Arch Intern Med 169:463–473. https://doi.org/10.1001/archinternmed.2008.603
Del Val D, Trottier M, Alperi A et al (2020) 18F-Fluorodeoxyglucose uptake pattern in noninfected transcatheter aortic valves. Circ Cardiovasc Imaging 13:e011749. https://doi.org/10.1161/CIRCIMAGING.120.011749
Acknowledgements
We would like to acknowledge the Infectious Endocarditis after TAVR International Registry Investigators for their substantial contribution to data collection and research (the list of investigators is available in the supplemental material). Dr Rodés-Cabau holds the Research Chair "Fondation Famille Jacques Larivière" for the Development of Structural Heart Disease Interventions. Dr. Panagides has received a research grant from the “Mediterranean Academic Research and Studies in Cardiology” association (MARS Cardio).
Funding
There was no specific funding for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Josep Rodés-Cabau has received institutional research grants from Edwards Lifesciences, Medtronic, and Boston Scientific. Vassili Panagides has received institutional research grants from Medtronic, Boston Scientific and Microport. C. Herrmann has received institutional research grants from Abbott, Boston Scientific, Edwards Lifesciences, and Medtronic and consulting fees from Edwards Lifesciences and Medtronic. Jan Malte Sinning reports speaker honoraria from Abbott, Boston Scientific, Edwards Lifesciences, and Medtronic and research grants from Boston Scientific, Edwards Lifesciences, and Medtronic, outside the submitted work. Kim Won-Keun reports personal fees from Boston Scientific, Edwards Lifesciences, Abbott, Medtronic, and Meril, outside the submitted work. Oliver Husser reports personal fees from Boston Scientific and payments from Abbott. Norman Mangner reports personal fees from Edwards Lifesciences, Medtronic, Biotronik, Novartis, Sanofi Genzyme, AstraZeneca, Pfizer, and Bayer, outside the submitted work. All other authors report no potential conflicts.
Rights and permissions
About this article
Cite this article
Panagides, V., Abdel-Wahab, M., Mangner, N. et al. Very early infective endocarditis after transcatheter aortic valve replacement. Clin Res Cardiol 111, 1087–1097 (2022). https://doi.org/10.1007/s00392-022-01998-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-022-01998-0